On December 2019, the world witnessed the outbreak of the novel coronavirus (SARS-CoV-2). By August 2021, more than 200 million people were infected and over 4.3 million individuals died.
For the first time ever, SHARP Corporation developed a device equipped with the patented Plasmacluster Technology. Under strict scrutiny and lab conditions, it was seen that there was more than 90% reduction on airborne and adhering novel coronavirus (SARS-CoV-2) including the Alpha variant (in adhering conditions).


“Disinfectants such as alcohol and detergents (surfactants) are well-known to be effective in reducing the risk of the virus on materials. However, for infection via aerosols (micro-droplets), there are few effective countermeasures such as a mask. SHARP demonstrated the effective inactivation of SARS-CoV-2 in airborne droplets by using Plasmacluster Technology. This will likely reduce the risk of infection in spaces like offices, homes, medical facilities and vehicles too”.
(Professor of National Research Center for the Control and Prevention of Infectious Diseases Nagasaki University)
The Mechanism for the Study.
- 1. Aerosolized virus passes through the test device from the generator
- 2. Virus is recovered post-exposure to Plasmacluster ions
- 3. Infectious virus titer is examined to calculate the virus density


“In order to prevent virus infection, it is important to maintain the environment at a relative humidity of about 60% by humidification, thereby preventing the human respiratory tract mucosa from drying out, and maintaining the protective function, and to suppress the infectivity of the virus. However, while an environment with a relative humidity of 60% reduces the number of suspended droplet particles, airborne droplet particles fall down and adhere to surfaces. Therefore, it is also necessary to consider the countermeasures against adhering viruses.
In this verification, Plasmacluster Technology significantly inactivates SARS-CoV-2 contained in adherent saliva in an environment with 60% humidity where the physiological protective function is maintained. The current result also shows its efficacy to the variant strain, which must be applied to new variants that will appear in the future.”
(Department of Microbiology-Shimane University Faculty of Medicine)
VERIFICATION OF AIRBORNE DROPLET PARTICLE DEPENDENCE ON HUMIDITY
In a simulated environment, SHARP examined the mobility of droplets at various humidity levels. The test confirms that at 60% humidity a higher volume of droplets adhered to the surface compared to humidity at 30%.
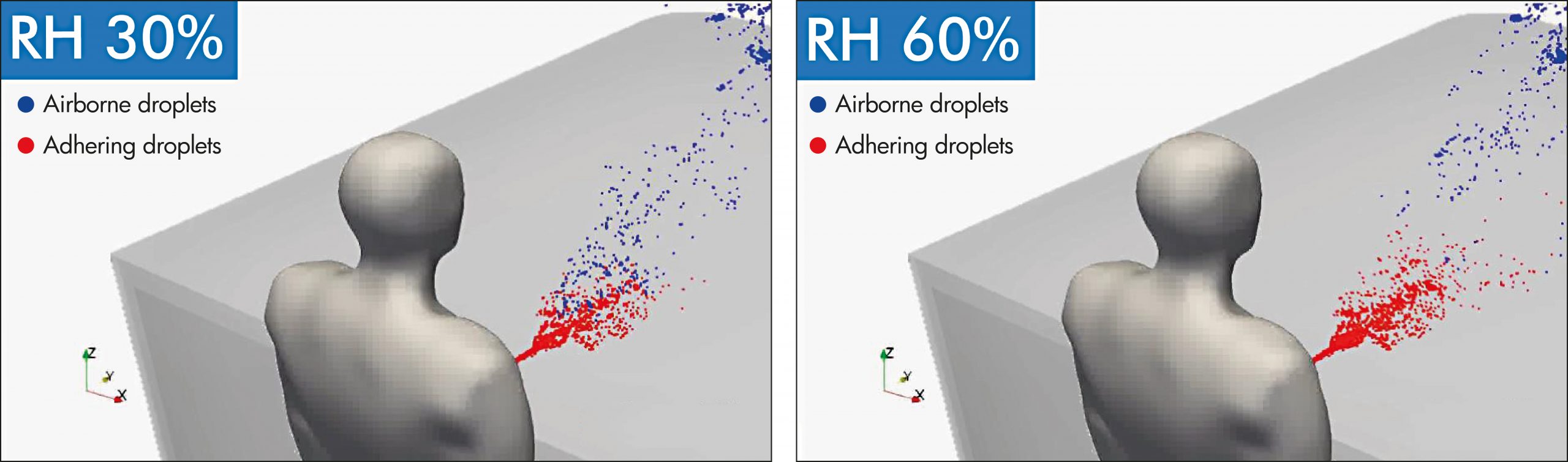
- Verifying organization: Department of Mechanical and System Engineering, Kyoto Institute of Technology
- Verification method: Simulation of droplet particles generated by coughing (while indoors)
- Simulation conditions: Temperature of 20°C; relative humidity of 30% and 60%
Results
1. It was confirmed that at 60% humidity fewer droplet particles remain suspended in the air.
2. As a result chances of inhaling viral droplets is lesser compared to when relative humidity is at 30%.
Effects of saliva on Viral Infectious Titer.
SHARP studied the survival rate of coronavirus in saliva. Compared to water the viscosity of saliva is 1.4-3.3 times higher.
- Testing organization: Department of Microbiology, Shimane University Faculty of Medicine
- Verification method: Compare infectious titers after allowing SARS-CoV-2 mixed with liquid medium and with saliva, respectively, to stand undisturbed for two hours
- Verification virus: Novel Coronavirus SARS-CoV-2
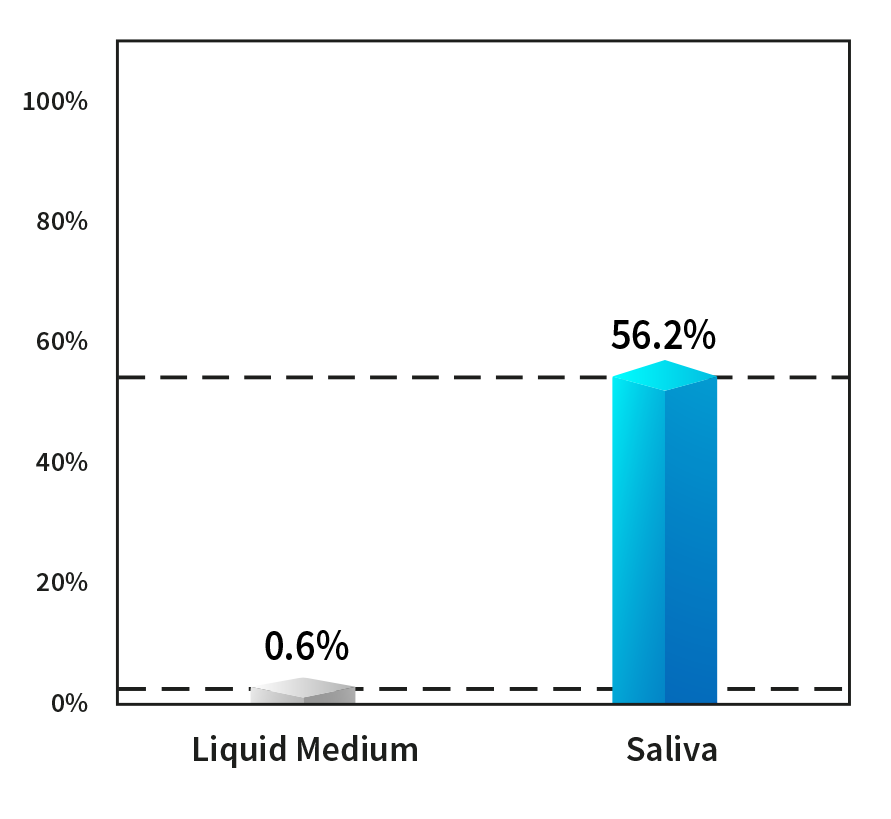
TEST CONDITIONS
Temperature of 20°C & Relative Humidity at 60%
(Liquid medium) D-MEM/Ham’s F-12 nutrient mixture
(Saliva) Saliva from 7 individuals (male and female)
Results
It was confirmed that the infectious titer in the liquid medium was less than 1%,
while the infectious titer in saliva remained at about 56%.
TEST CONDITIONS
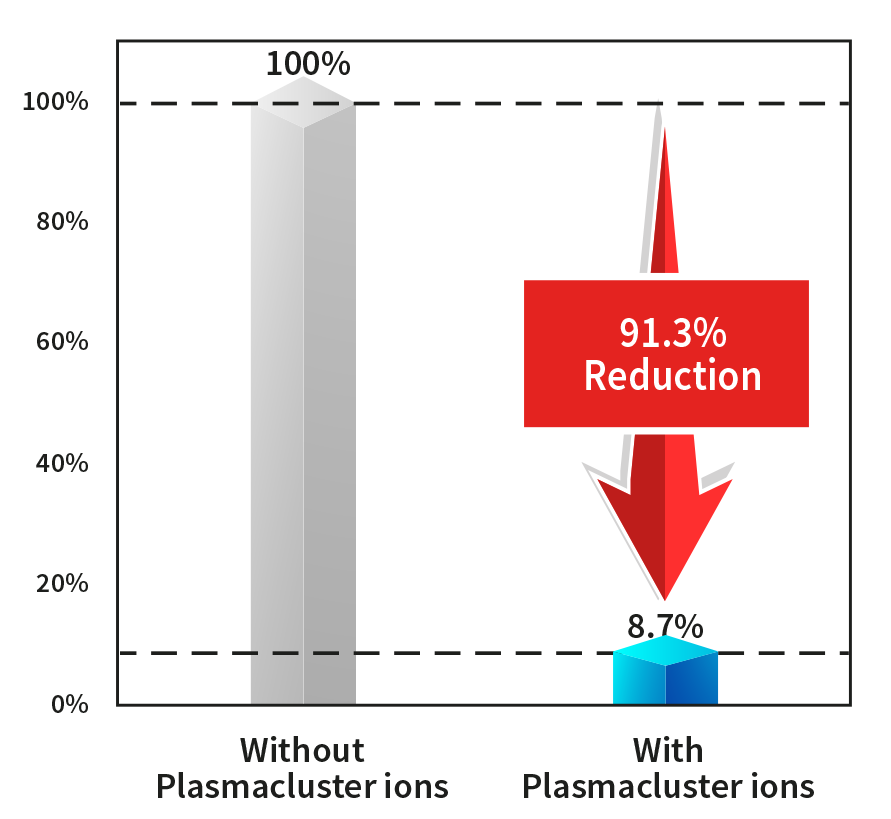
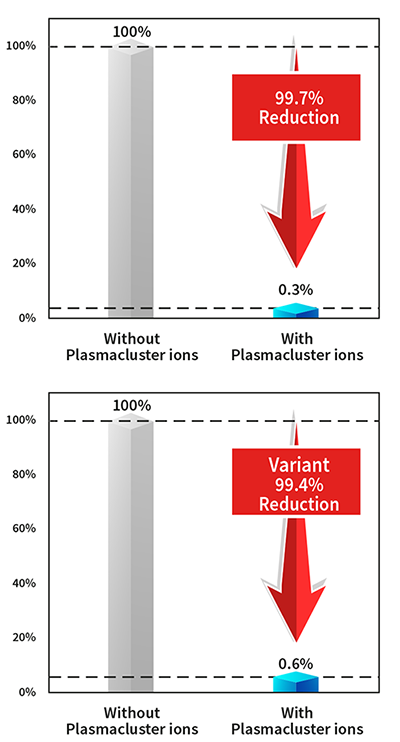
Verification of Plasmacluster Technology’s Efficacy on Adhering Coronavirus in Human Saliva
- Testing organization: Department of Microbiology, Shimane University- Faculty of Medicine
- Verification apparatus: Adherent virus test device equipped with Plasmacluster Technology
- Plasmacluster ion density: Approx. 600,000/cm3 (ion exposure distance: 10 cm)
- Test space: Approx. 38 liters
- Test conditions: Approx. temperature- 20°C; relative humidity- 60%
- Verification virus: Novel Coronavirus SARS-CoV-2 and variant strain (Alpha variant)
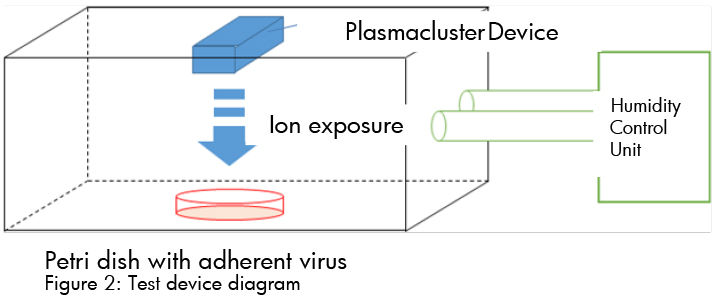
TEST PROCEDURE
- Saliva mixed with virus solution
- 50 micro liters virus solution exposed to Plasmacluster ions for 2 hrs.
- Viral infectious titer (TCID50/ml) from the recovered virus solution is calculated using TCID50 assay
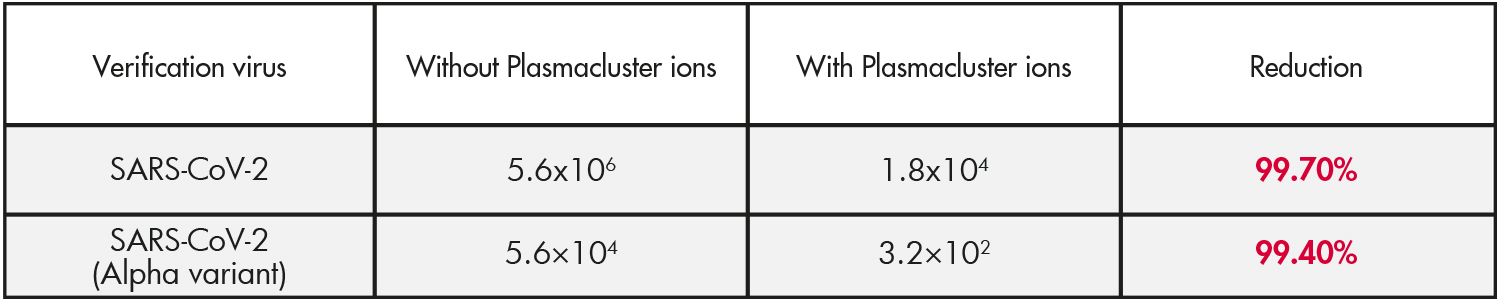
Results
Infection titer (TCID50/ml) and reduction rate of new coronavirus contained in adherent saliva